Clinical Center News
New DTM machine reduces contamination and saves labor in cell expansion
The ease and security of automation is coming to bone marrow stromal cell expansion in a new machine in the Clinical Center’s Department of Transfusion Medicine (DTM). Reducing the current manual method guarantees less contamination and frees staff time for other procedures.
From the Trans-NIH Bone Marrow Stromal Cell Transplantation Center came a partnership with technology developer CaridianBCT to install the cell expansion system, the prototype for such a machine.
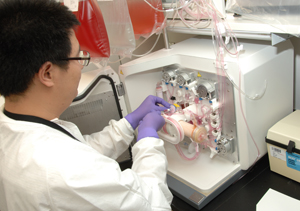 |
Staff scientist Jiaqiang Ren samples a milliliter of cell culture media from the cell expansion system once or twice a day to measure glucose and lactate levels, parameters used to estimate cell expansion and, in turn, regulate how fast to feed the cells. The cell expansion system—the cylinder that Ren pulls aspirate from above—replicates the desired amount of bone marrow stromal cells in an average of five to seven days.
|
The center (a joint initiative by the CC, NCI, NIAID, NIBIB, NIAMS, NIDCR, and NINDS) was created in 2008 to facilitate the use of clinical grade bone marrow stromal cells prepared using procedures known to maintain their biological activities and to assist investigators in the preparation of protocols that utilize such cells. The trans-NIH group is co-coordinated by Dr. Pamela G. Robey of the National Institute of Dental and Craniofacial Research and Dr. Harvey G. Klein, chief of the DTM.
Bone marrow stromal cells (also called mesenchymal stem cells) form the supportive structure in which the cells that produce human blood reside and play a part in the repair of tissue and bone formation. Patients with auto-immune disorders, acute host-versus-graft disease, and need for bone formation after surgery benefit from the transfusion or direct transplantation of these cells.
Dr. Keith A. Horvath, director of the National Heart, Lung, and Blood Institute’s Cardiothoracic Surgery Research Program and chief of cardiac surgery at the NIH Heart Center at Suburban Hospital, plans to inject these cells into his patients with ischemic heart disease for quicker recovery of damaged tissue, noted Dr. David Stroncek, chief of the DTM’s Cell Processing Section.
In the traditional expansion method, cell processing laboratory staff take a piece of bone or bone marrow aspirate, a sample of the liquid bone marrow portion, and replicate the stromal cells by growing them in flasks and transferring to larger flasks as they grow in number. Staff must monitor the cells’ development and move them by hand.
“This process doesn’t lend itself well to a clinical cell processing environment because the system is open. Every time cells are transferred from one flask to another they could become contaminated with bacteria or fungus,” Stroncek said.
The new cell expansion system produces cells in a sealed container using bioreactor technology, which circulates fluid through cartridges and automatically loads the cells into the cartridge, feeds the cells, and harvests the cells. CaridianBCT’s system also grows cells at a higher density in the same volume of fluid, saving expensive media.
“Having a technology that enables the production of large quantities of cells in a reproducible, robust, and closed-system manner is a key to being able to move forward with the clinical validation and subsequent implementation of cellular therapies,” said Klein.
Samples will come from patients or healthy volunteers. While any healthy person can donate marrow to produce bone marrow stromal cells, Stroncek listed people who have volunteered to donate bone marrow or peripheral blood stem cells for transplantation into compatible relatives or strangers as a potential base for recruitment.
Until the machine’s output is verified as comparable to the expanded stromal cells created through traditional methods, the DTM will continue to make cells for patients using a more traditional flask/cell factory method. Within six to nine months, cells produced using the CaridianBCT Cell Expansion System should be ready for treatments, Stroncek said.
Back to Top
NIH Center focuses on imaging of infectious disease
 |
External view of the NIAID Integrated Research Facility in Frederick, Md.
Image courtesy of Smith Carter ©2009
|
Looking at common illnesses like influenza and rare conditions such as hemorrhagic fevers, Clinical Center imaging specialists have begun a partnership to learn more about the presentation and characteristics of various infectious diseases.
The CC Radiology and Imaging Sciences group has teamed with the National Institute of Allergy and Infectious Diseases to establish the new Center for Infectious Disease Imaging, which will use state-of-the-art imaging methods to understand, diagnose, and aid the medical management of public health threats such as influenza, severe acute respiratory syndrome (SARS), poxviruses, and other emerging pathogens.
“The radiology and NIAID partnership will form an image-based strategy to understand infectious diseases and translate that understanding into applications for diagnosis and staging and for monitoring response to therapy,” said Dr. Daniel Mollura, a staff radiologist and scientist who recently came to the CC from The Johns Hopkins Hospital to work with the Center for Infectious Disease Imaging.
Research will take place at the newly constructed NIAID Integrated Research Facility (IRF) at Fort Detrick in Frederick, Md. The IRF is a state-of-the-art research laboratory designed to conduct research in biological safety laboratory level 4 conditions—the highest possible rating.
“The IRF was designed with unique architecture and engineering to enable secure, safe, and specialized medical imaging, including magnetic resonance imaging, positron emission tomography, computed tomography, ultrasound, and single positron emission computed tomography,” said IRF Director Dr. Peter B. Jahrling.
The unique IRF environment and multidisciplinary Center for Infectious Disease Imaging team will enable characterization of infectious diseases that has never been done before with advanced imaging methods. Such understanding of how influenza, SARS, and other infectious diseases present and travel through the human body will be the first step in the field’s translational research mission.
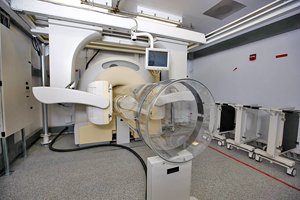 |
Scientists at the Center for Infectious Disease Imaging will use advanced technologies such as this single positron emission computed tomography (SPECT CT) machine to identify functional changes inside the body.
Image courtesy of NIAID
|
“The center will have the tools available to allow discoveries in infectious disease imaging to be translated to the care and treatment of patients,” noted Dr. David A. Bluemke, director of Radiology and Imaging Sciences and acting director of the Center for Infectious Disease Imaging.
Advanced imaging will help clinician scientists learn more about how infections present, allowing for quicker disease detection. Imaging pathogen movement throughout the body at systemic and cellular levels will also help scientists determine optimal therapies and tracking strategies, said Mollura.
“This effort will be critical for understanding the pathophysiology of infectious diseases that are potential threats to public health,” he said.
Back to Top
NINDS Nueroimmunology Branch chief becomes emeritus
Long-time chief of the National Institute on Neurological Disorders and Stroke’s Neuroimmunology Branch, Dr. Henry F. McFarland retired and was appointed scientist emeritus in late July.
Many coworkers and old friends gathered for a reception on July 29 to wish the leading multiple sclerosis researcher well in the next step in his more-than-30-year NIH career.
“What you’ve done is extraordinary—not just the science, but the friendships that you have made,” said Dr. Steven Jacobson, chief of the NINDS Neuroimmunology Branch’s Viral Immunology Section. “People can succeed you, but no one can replace you.”
McFarland earned his bachelor’s degree from the University of Arizona in 1962 then his medical degree from the University of Colorado in 1966. After residency in neurology at Thomas Jefferson Medical College in Philadelphia, he completed two NIH special fellowships—one in neurovirology and neurology at Johns Hopkins University School of Medicine, and one in immunology at University College in London.
McFarland first came to the Bethesda campus in 1975 as deputy chief of the Neuroimmunology Branch of the then National Institute of Neurological and Communicative Disorders and Stroke. He has been chief of that branch since 1992 (acting for the first two years) and chief of the Neurological Disease Section since 1999. McFarland also served as director of NINDS’ Clinical Neurosciences Program from 2001 to 2007.
His awards include the USPHS 1996 Physician Clinical Researcher of the Year, 2000 and 2009 NIH Director’s Awards, and the Charcot Award in 2003 from the International Multiple Sclerosis Federation for lifetime achievement in multiple sclerosis research.
 |
Dr. Henry F. McFarland bid “see you soon” to NINDS coworkers and NIH friends at a reception in his honor on July 29. The long-time chief of the institute’s Neuroimmunology Branch became scientist emeritus earlier this summer.
|
In addition to his numerous affiliations with multiple sclerosis committees and societies over the years, McFarland was on the Advisory Committee for Clinical research and is a member of the NIH Inflammatory and Autoimmunity Working Group on Biomarkers.
At the reception in his honor, McFarland reflected on his love for the work he’s done for three decades and for the people he’s done it with. “We enjoy coming to work. We have a mission,” he said. “That mission and the people together, it’s been just wonderful.”
The Neuroimmunology Branch is doing much to remember their leader, with a meeting on multiple sclerosis in the District of Columbia planned for later this year and the branch’s new conference room named for McFarland.
The Clinical Transition to Independence Award will also bear his name, announced NINDS clinical director Dr. Mary Kay Floeter.
Back to Top
Hospitality Services reorganizes to deliver quality customer service
Seven months after a reorganization of the Office of Hospitality Services, the group has begun to implement changes that improve the patient experience—from the front door to the bedside.
Previously under the Office of Organizational Development, Hospitality Services branched off in early 2009 with a broader goal of helping the Clinical Center deliver outstanding customer service consistently and seamlessly across the front line. The office now reports directly to Maureen Gormley, CC chief operating officer who continues to focus on improvement of service excellence across the CC.
The reorganized Hospitality Services continues to be led by CAPT Denise Ford. Responsibilities include customer service representatives at hospitality desks and the patient entrance at West Drive, staff at reception desks in front-line departments, patient ambassador volunteers, and the patient escort staff. In an effort to establish consummate service throughout the patient experience, Ford established a partnership with Gina Mattia, the chief medical technologist of phlebotomy services, so that shared customer service standards were established between phlebotomy and the other front-line services. This service excellence team now spans from the north lobby’s hospitality desk to the front desks of phlebotomy, the EKG/heart station, radiology, and nuclear medicine.
“We’re all committed to the same standards,” Ford said.
One of the first actions Hospitality Services took was adding a critical element on service excellence standards to employees’ Performance Management Appraisal Plan. This step adds accountability, Ford explained, and staff will be evaluated through supervisor observation and patient feedback and surveys.
Managers should model for their staff what constitutes good customer service—a warm greeting and welcoming body language make a difference. Employees should be proactive in offering assistance, Ford said.
“You know how you want to feel when you go somewhere new—put those eyes on and you’ll recognize more opportunities to help,” said Yasmin Coates, Hospitality Services’ events program coordinator.
Small changes have made significant differences. Hospitality Services now handles scheduling for radiology, nuclear medicine, and pre-anesthesia services. The addition of a cancel option to the automated schedule-by-phone system has allowed appointments to be quickly cancelled if a patient’s schedule changes. Since approximately 20 percent of patients do not arrive for appointments, this approach has the potential to substantially reduce the patient backlog, allowing better access for other patients. Over 100 additional appointment slots were freed up for Radiology and Imaging Sciences procedures in the first month that the cancel option system was in place.
Hospitality Services also refurbished outdated signage instructing patients on how to contact the radiology technicians for after-hours services, thereby decreasing their wait time. “It’s the little things that make the experience easier and smoother for the patients,” said Ford.
Next steps include removing the glass barrier between receptionists and visitors at the radiology check-in desk. A patient survey is being conducted in the department to identify other areas for improvement. These changes came out of the CC’s Service Excellence Committee co-chaired by Gormley and Radiology and Imaging Sciences Director Dr. David Bluemke.
Back to Top
CC staff receive NIH Director’s Awards
The following Clinical Center staff were honored with an NIH Director’s Award at a ceremony on July 29.
Denise Ford: chief, Hospitality Services: Mentoring—for outstanding contributions as a mentor, teacher, and leader of employees and volunteers in support of excellent customer service across the CC
Dr. James Shelhamer: deputy chief, Critical Care Medicine Department: Scientific/Medical—for extraordinary contributions to the understanding of pulmonary physiology
Dr. Robert Wesley: chief, Biostatistics Service: Scientific/Medical—for extraordinary contributions to the design and analysis of clinical research projects
Dr. David Henderson: deputy director for clinical care: Administrative—for use of novel leadership strategies to facilitate high quality clinical research and for the application of epidemiological methods to enhance patient safety
Karen Kaczorowski: deputy chief nurse officer, Ambulatory Care Services: Administrative—for outstanding work on strategic initiatives
CC Patient Travel Team (Maureen Gormley, John McKeeby, Dr. Adrienne Farrar, Kim Jarema, Karen Kaczorowski, Sydney Jones, Bridget Moore): for exceptional teamwork in creating a new administrative infrastructure to implement a uniform and equitable NIH patient travel policy
NIDA RTX Development Project Members (Dr. Gopal Potti, Dr. George Grimes, Judith Starling, Dr. Andrew Mannes): for advancing resiniferatoxin to a successful IND approval, enabling its evaluation in clinical trials as a treatment for intractable pain.
Walter Jones: lead of diversity management and minority outreach, Office of Clinical Research Training and Medical Education: as a member of the NIH Health Disparities Summit group for contributions in planning and coordinating the first NIH Summit: The Science of Eliminating Health Disparities
Back to Top
First class of neuropsychiatry nursing fellows graduates
 |
|
Dr. Clare Hastings (left), Nursing and Patient Care Services chief, helped Kim Cox (second from left) and Julie Kohn (right)—the women behind the scenes of the Neuropsychiatry Nurse Fellowship Program—graduate eight participants on August 14. Completing the program were: (back row, from left) Marina Budzynski, Jacqueline M. Goodson, Alisha Holden, Salieu Mansaray, Kathryn J. Prescott, and (front row, from left) Maria Gabriela Tietcheu, Erin Nicole Torres, and Jerome Glassman.
|
The Clinical Center Nursing and Patient Care Services Neuropsychiatry Nurse Fellowship Program graduated its first class at a ceremony on August 14.
Program coordinator Julie Kohn and Dr. Clare Hastings, chief of Nursing and Patient Care Services, presented eight nurses with a certificate of completion and smiles of pride and encouragement.
Marina Budzynski, Jerome Glassman, Jacqueline M. Goodson, Alisha Holden, Salieu Mansaray, Kathryn J. Prescott, Maria Gabriela Tietcheu, and Erin Nicole Torres graduated the year-long training in front of an assembled group of family and coworkers.
“You now have a foundation, a framework to practice,” said Kim Cox, psychiatric-mental health clinical nurse specialist with the CC Nursing and Patient Care Services’ Research and Practice Development Service, who helped run the program with Kohn.
Offering congratulations and well wishes were leaders of the programs the graduating nurses serve—Dr. Maryland Pao, clinical director of the National Institute of Mental Health; Dr. David Ted George, chief of the National Institute on Alcohol Abuse and Alcoholism’s Section of Clinical Assessment and Treatment Evaluation; and Bruce Steakley, nurse manager of the Child and Adolescent Psychiatric-Mental Health Research Unit and the Psychiatric-Mental Health Outpatient Research Program.
George instructed the class to overcome the “boring routines, the busyness, the hierarchies” by serving a greater purpose. “We all deal with everyday-ness,” he said. “Get a mission. Have a focus—something you’re driving toward. Number one should be patient care.”
Back to Top
BTRIS lead will hold town hall meeting as system implements phase II
Phase II of the Biomedical Translational Research Information System (BTRIS), launching this month, will allow NIH researchers to access and analyze de-identified data from NIH intramural protocols. This aspect of BTRIS is designed to develop new research ideas and to do population-based summaries or detailed queries.
BTRIS, which went live on July 30, provides users with advanced search, filtering, and aggregation methods to create data sets to support ongoing studies. The second push will supply users with subject’s de-identified data from the Clinical Center’s Clinical Research Information System, the Clinical Research Information Management System of the National Institute of Allergy and Infectious Diseases, and the National Institute on Alcohol Abuse and Alcoholism database. Phase I offered clinical investigators the opportunity to search through and sort data from patients only on their own protocols.
The following reports are available in BTRIS 1.0: Institutional Review Board Inclusion Enrollment Report, patient demographics, patient lists, laboratory and microbiology results, vital signs, medication orders, and administration. Additional reports will become available through the fall. Future versions of BTRIS will contain additional historical and clinical data back to 1976, images, and subject data from other institute and center’s systems such as that of the National Cancer Institute.
To assist staff in navigating the new system, the BTRIS User-Support Center is open in the Hatfield Building’s 4-2480 (to the right of the Medical Board Room) from 8:30 am to 5:00 pm, Monday through Friday, through October 30. The team also provides self-paced, computer-based training modules for those who want to learn on their own.
To speak on the development and implementation of and hopes for BTRIS and to answer questions from current and potential users, Dr. Jim Cimino, chief of the CC Laboratory for Informatics Development, will give the concluding presentation in the BTRIS Lecture Series that welcomed leading figures in the study and use of translational information systems from academic centers across the United States. The September 15 lecture will take place at 2 pm in the Lipsett Amphitheater.
For more information on BTRIS, visit http://btris.nih.gov.
Back to Top
CC Green Team rolls out environmental sustainability initiatives
By: Nicole Martino
The Clinical Center has recently joined the ranks of hospitals across the country working toward a healthier environment with the creation of the CC Green Team.
“Hospitals in general use tremendous amounts of resources and energy, and there are simple things that we can do to make a big impact,” said Dave Folio, head of the CC Nutrition Department and CC Green Team leader. “At home, you recycle, you shut off the lights when you leave a room. We want to instill that same awareness here at work.”
The CC Green Team looks at how the center conducts its day-to-day activities and recommends earth-friendly initiatives to support the team’s goals of increasing recycling, decreasing paper consumption, and decreasing the amount of electricity used by the CC. The NIH Bethesda campus uses approximately $65.5 million worth of electricity per year, and about 10 percent of that is used by the CC, according to the Office of Resource Facilities.
A collaboration with the NIH Environmental Management System, the CC Green Team was formed to comply with an executive order to strengthen federal environmental and energy management and because “it was simply the right thing to do,” Folio said.
Evidence of the Green Team’s efforts can be found throughout the building—from the hallway recycling containers brimming with plastic and paper to the reusable plates on which CC patients receive their food.
 |
Linda Collins, Nutrition Department program support, assists with implementation of the Green Team’s desk-side recycling bin initiative as CC staff member Kristy Suhr picks up her bins.
|
One recent step to promote a greener CC is the desk-side recycling bin initiative. Staff can request two small, blue bins—one for commingled (cans and bottles) and one for mixed paper—for their desk side. The Green Team has already distributed more than 700 bins and will evaluate the progress of this initiative by measuring the weight of commingled and paper recyclables over time, said Folio.
Employees and visitors can help serve the Green Team mission. When leaving a room for more than a few minutes, turn off the lights. This small step can save roughly three cents an hour, which, considering the CC’s many rooms, can really add up. Employees should recycle ink and toner cartridges in the bins throughout the CC, especially because for each cartridge recycled, $1 goes to NIH charities like Friends of the Clinical Center and The Children’s Inn. Employees can also help reduce paper consumption by decreasing type size to save toner and copying and printing on both sides of paper if possible.
The Green Team would like to remind staff to dispose of materials in the proper receptacles—paper with sensitive information should be shredded and not placed in a recycling bin, and regular waste and recyclable materials are not to go in burn boxes.
For more information about greening initiatives at the CC or about NIH’s commitment to the environment, visit NIH Environmental Management Systems at http://www.nems.nih.gov/.
Back to Top
New barcode system Phase II launches
The updated barcodes on inpatient wristbands and patient labels will be affixed to outpatient’s extended visitor badges and used for outpatient specimen collection beginning early this fall. A station at the Clinical Center’s B1 entrance will provide patients already possessing an extended visitor badge a barcode to affix to the badge noting their medical record number. Those who wish not to apply for an extended visitor badge will be issued a wallet id card showing only their name, picture, and barcode. New patients will automatically receive a wallet id card and an application for an extended visitor badge. New extended visitor badges and wallet id cards will have the holder’s medical record number encoded in the barcode printed directly on the badge.
The barcode change minimizes human error in medicine and blood administration and in specimen collection by requiring staff to scan the patient’s barcode at various points while drawing blood or giving medication, for example. This multiple identification-check process is possible due to a workstation on wheels in each inpatient room and at phlebotomy services.
Patients should ask a Hospitality Services representative or member of their research team how to get the barcode label or wallet id card. New patients will be instructed on the process in Admissions.
Staff will be required to attend training on how to use the barcode devices. Look for an e-mail with instructions in the coming weeks.
Back to Top
Flu vaccine mandatory for those with patient contact
 |
For the second year, NIH patient-care staff will be required to receive or formally decline the influenza vaccine. Available free of charge, the vaccine will be offered starting in October.
This year NIH may be able to offer vaccines to contractors who have contact with patients. Additional information about this new program will be available within the next month. Such contact employees will need to be vaccinated or complete a form stating their contraindication or reason for declination. Staff who fall under the requirement will be notified by e-mail.
Protecting oneself against the highly communicative virus is a testament to dedication to patient care and a recommendation of The Joint Commission. Still, many staff choose not to protect themselves and those around them, including the Clinical Center’s patients (many of whom are immunosuppressed), family, and coworkers.
NIH offers many opportunities to comply with the mandate, approved by the Medical Executive Committee in February 2008. The Traveling Flu Show will bring nurse vaccinators to CC inpatient units and outpatient clinics the week of October 5 to 16. Staff should watch for flyers publicizing the date and time of a visit to their work area.
A clinic open to all who present an NIH employee (non-contractor, unless required) badge will offer the influenza vaccine on the Hatfield Building’s seventh floor the weeks of October 19 to 23 and November 2 to 6. The clinics are organized by last name, and all employees will have two opportunities to visit the clinic. Vaccinators will also hold sessions at off-campus locations.
The influenza vaccine for the 2009-2010 season contains the following strains recommended by the Food and Drug Administration’s Vaccines and Related Biological Products Advisory Committee: A/Brisbane/59/2007 (H1N1)-like, A/Brisbane/10/2007 (H3N2)-like, and B/Brisbane/60/2008-like virus. This vaccine will not prevent against the novel H1N1 agent that was introduced from Mexico this spring, but will prevent infection from the other seasonal strains that have been circulating. Several additional vaccines are in production that will prevent infection with the novel H1N1 agent. The novel H1N1 vaccine should become available later in the fall. As infection with influenza can be life-threatening for many of our patients, immunization with both vaccines will be required for all individuals who have contact with our patients. When this issue of CC News went to print, NIH expected to purchase a number of H1N1 vaccines, but did not know yet an amount or date of delivery. The distribution plan for the novel H1N1 vaccine is contingent on that information.
A full vaccination schedule will run in the October issue of CC News and will be posted at http://foiltheflu.nih.gov. For more information, call the CC Hospital Epidemiology Service at 301-496-2209.
Back to Top
New diagnostic imaging devices to automatically record radiation exposure
Imaging tests are necessary for disease detection and monitoring, but the radiation that such machinery emits may have lasting effects. To further safeguard clinical research patients, the Clinical Center’s Radiology and Imaging Sciences has taken a significant step.
The next generation of computed tomography (CT) and positron emission tomography (PET)/CT equipment purchased by the CC will now be required to routinely record radiation dose exposure in a patient’s electronic medical record
“When a hospital or clinic patient receives a medication or a treatment, it is routinely recorded,” said CC Director Dr. John I. Gallin. “The Clinical Center’s approach is an important first step in making it possible to more easily document and track information about a patient’s exposure to radiation.”
About 25,000 CT and 1,250 PET/CT scans are performed each year on the CC’s five CT scanners and two PET/CT scanners. Many patients must have these tests numerous times to track the progression or regression of their disease. Today’s electronic radiology information systems in hospitals generally do not collect or report radiation exposures.
It is known that high radiation doses have the potential to cause cancer, but the effect of low doses, such as from medical-imaging tests, is not yet understood. The ability to keep track of an individual’s exposure to radiation through routine imaging tests is needed so that researchers can begin to determine if these exposures pose a health risk. Ultimately, radiation dosage could become a standard element of a universal electronic medical record used to assess radiation risk from life-long medical testing, CC radiologists said.
“The National Council on Radiation Protection and Measurements reported recently that Americans received seven times more radiation exposure from medical tests in 2006 than was the case in the 1980s,” said Dr. Ronald Neumann, chief of nuclear medicine and deputy associate director for imaging sciences. “CT and cardiac nuclear medicine studies accounted for much of this increased medical radiation exposure.”
Radiology and Imaging Sciences Director Dr. David A. Bluemke announced, “The NIH Clinical Center also will require that newly purchased equipment allows patients to record their radiation dose exposure in their own personal health record.”
Online resources to help individuals organize their health information are becoming more prevalent. Currently, patients can easily receive their diagnostic imaging studies records on CD-ROM, Bluemke said. The CC will work with imaging equipment vendors to develop software tools to extract the examination type, date, and radiation dose exposure from the CD-ROM, for uploading to a personal health record.
Back to Top
Sibling piano prodigies perform in CC atrium
Sister and brother prodigies, Olivia and Jeffrey Ly, 13 and 14 respectfully, played a duet piano concert on the Clinical Center’s Steinway grand piano in the Hatfield Building’s atrium on August 20. The pair were accompanied by their long-time instructor, Dr. Bella E. Oster, professor of piano performance at the European Academy of Music and Art, Inc. The siblings attacked the ivories with passion on arrangements by Strauss, Gershwin, and Brahms.
Back to Top
“Reliable” HHS badge ushers in era of stricter identity proofing
In accordance with Homeland Security Presidential Directive 12—requiring all federal agencies to use a “reliable, commonly accepted form of government identification that will enhance security, increase government efficiency, reduce identity fraud and protect personal privacy”—all NIH employees, contractors, and affiliates (fellows, guest researchers, and special volunteers) will be issued a new badge.
Also called PIV Card or smart card, the Department of Health and Human Services badge will be reliable because its holder must undergo a strict personal identity verification.
Issuance will begin this month and continue through June 2010. Throughout the badging process employees, contractors, and affiliates will receive detailed instructions by e-mail on how to proceed—notifying them when to make an enrollment or issuance appointment, and highlighting ‘next steps’ and how to get help. The NIH Enterprise Directory team will “force” the renewal of all badges, even those that show an expiration date that occurs well into the future.
If a badge holder already has a background investigation on file, no additional forms must be completed. The individual will still have to be photographed and have two fingerprints captured.
Individuals without the appropriate background investigation on file (usually new federal employees, but also a sizeable percentage of current employees) will have all 10 fingerprints captured and must fill out a questionnaire on their personal history.
Once the enrollment process is complete, an individual will be notified via e-mail to schedule an appointment to pick up the new HHS badge at an Issuance Work Station. The new badge will admit the holder to all areas that s/he had previous access to with his/her NIH badge.
For a complete description of the new HHS badge and the personal identity verification process, visit http://idbadge.nih.gov.
Back to Top
News Briefs
Renowned poet to speak in Masur Auditorium
Maya Angelou, notable poet, novelist, educator, actress, and more, on September 23 will present the annual J. Edward Rall Cultural Lecture in the Clinical Center’s Masur Auditorium at 3pm. The National Center on Minority Health Disparities, in partnership with the Office of Intramural Research, will host Angelou as part of the Wednesday Afternoon Lecture Series. She is most recognized as the author of I Know Why the Caged Bird Sings.
Seating is on a first come, first served basis. Overflow seating will be available in Lipsett Amphitheater. Visit http://wals.od.nih.gov/ for more information.
Piano Concert Series performances
September 16, 2009
Noon to 1 pm, Hatfield atrium
H. David Meyers and Friends (Chamber Soloists of Washington) will perform an eclectic program of classical and contemporary works. Meyers, an oboist who has soloed with members of the National Symphony, will be joined by musicians from many local, renowned orchestral groups.
October 1, 2009
Noon to 1 pm, Hatfield atrium
Members of the Ars Nova Chamber Orchestra will perform works by Marcello, Schumann, Brahms and Mozart. Performers include Marcus L. Jordan, clarinet; Sayaka Kanno, piano; Raea Jean Leinster, viola; and Wenyin Chan, piano.
Back to Top
Upcoming Events
All lectures will be videocast at http://videocast.nih.gov.
September 2, 2009
CC Grand Rounds
Lipsett Amphitheater, 12 noon
Update on Mood Disorders: International Patterns and Impact of Mood Disorders
Kathleen R. Merikangas, PhD
Chief, Genetic Epidemiology Research Branch, NIMH
An Update on Drug Discovery and Treatment Biomarker Development in Mood Disorders at NIMH
Carlos A. Zarate, Jr, MD
Chief, Mood Disorders Research Unit, Mood and Anxiety Disorders Program, NIMH
September 9, 2009
CC Grand Rounds
Lipsett Amphitheater, 12 noon
Contemporary Clinical Medicine: Great Teachers
Treatment of Early Breast Cancer
Norman Wolmark, MD
Chairman, National Surgical Adjuvant Breast and Bowel Project
Professor and Chairman, Department of Human Oncology, Drexel University College of Medicine
Chairman, Department of Human Oncology, Allegheny General Hospital
September 16, 2009
CC Grand Rounds
Lipsett Amphitheater, 12 noon
Epidemiology and Immunologic Risk Factors for Age-Related Macular Degeneration
Emily Chew, MD
Deputy Director, Division of Epidemiology and Clinical Research, NEI
New Evidence that Age-Related Macular Degeneration is an Immunologic Disease
Robert Nussenblatt, MD, MPH
Chief, Laboratory of Immunology, NEI
Acting Scientific Director, NCCAM
Wednesday Afternoon Lecture Series
Masur Auditorium, 3 pm
Targeting Chk1 in Breast Cancer
Helen Piwnica-Worms, PhD
Gerty T. Cori Professor of Cell Biology and Physiology,
Washington University in St. Louis
HHMI Investigator
September 23, 2009
CC Grand Rounds
Lipsett Amphitheater, 12 noon
Deep Venous Thrombosis: Old Problems, New Options
Jay Lozier, MD, PhD
Staff Clinician, Hematology Service, Department of Laboratory Medicine, CC
Richard Chang, MD,
Chief, Endocrine and Venous Services Section, Interventional Radiology Section, Radiology and Imaging Sciences, CC
Wednesday Afternoon Lecture Series
Masur Auditorium, 3 pm
J. Edward Rall Cultural Lecture
Maya Angelou
Reynolds Professor of American Studies, Wake Forest University
September 30, 2009
CC Grand Rounds
Lipsett Amphitheater, 12 noon
Validation of Advanced Cardiac Diagnostic Imaging and Specific Clinical Indications
Marcus Y. Chen, MD
Staff Clinician, Translational Medicine Branch, Laboratory of Cardiac Energetics, NHLBI
Coronary Artery Disease: Should We Treat Ischemia, Stenosis, or Atherosclerosis?
Andrew E. Arai, MD
Senior Investigator, Translational Medicine Branch, Laboratory of Cardiac Energetics NHLBI
Wednesday Afternoon Lecture Series
Masur Auditorium, 3 pm
Cholesterol, HIV and AIDS
James Hildreth, MD, PhD
Director, Center for AIDS Health Disparities Research
Associate Director, Vanderbilt-Meharry Center for AIDS Research
Professor, Meharry Medical College
Back to Top
This page last updated on 12/14/2017


 The information on this page is archived and provided for reference purposes only.
The information on this page is archived and provided for reference purposes only.